- Details
Our Patient Education Events are in-person, full-day opportunities to learn, led by top acoustic neuroma specialists. Patients hear about treatment options, get clear explanations of different procedures, and learn what to expect during recovery. Together with fellow patients, attendees reach a keen understanding of what their acoustic neuroma journey could look like.
*****2026 PATIENT EDUCATION EVENTS*****

Virtual Patient Education Event
February 7, 2026 (rescheduled)

In-Person Patient Education Event
Mayo in Florida - Jacksonville
March 7, 2026

In-Person Patient Education Event
Los Angeles, CA
October 3, 2026
- Details
The Great Brain Freeze
 The Great Brain Freeze is the Acoustic Neuroma Association's signature winter fundraising theme encouraging friends, family and members of your local community to join you in taking an icy plunge to raise awareness and funds for the Acoustic Neuroma Association.
The Great Brain Freeze is the Acoustic Neuroma Association's signature winter fundraising theme encouraging friends, family and members of your local community to join you in taking an icy plunge to raise awareness and funds for the Acoustic Neuroma Association.
If brraving an arctic dip is not your cup of tea or your winter weather is always sunny, consider a pool party, ice cream social or group snowball fight. The possibilities to keep it cool for your fundraising are endless!
The Great Brain Freeze is the brainchild of Neil Donnenfeld, ANA Vice-President and AN patient. Neil's inaugural plunge into the chilly Atlantic Ocean took place on February 4, 2018 at Fisherman's Beach in Swampscott Massachusetts. Watch his story:
To get started on Your Great Brain Freeze:
- Talk with your doctor to ensure you can participate in this type of activity.
- Email our development team to let them know of your participation.
- Review our Event Tips and Guidelines and complete and return the Event Information Submission Form.
- Download our helpful templates and The Great Brain Freeze logo (1-color and 2-color formats) to promote your event.
- Details
Clint Sarion
Clint Sarion - The Smiling Ninja and an AN Warrior


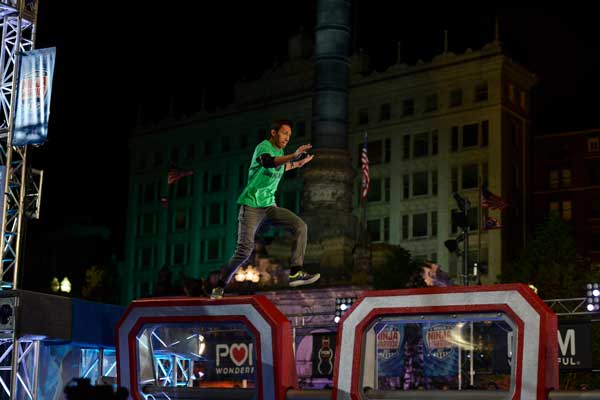
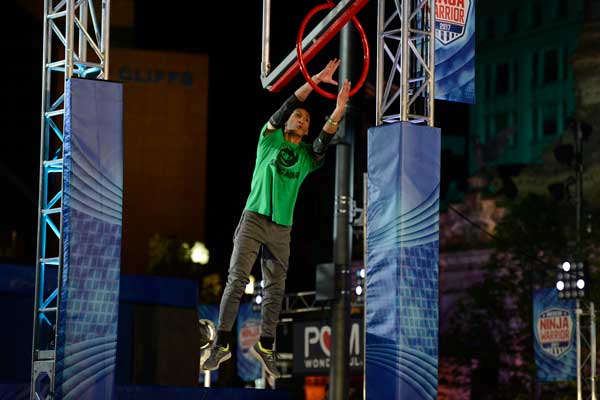
AMERICAN NINJA WARRIOR --"Cleveland Qualifier" -- Pictured: Clint Sarion -- (Photos by: Duane Prokop/NBC)
Read Clint's story https://m.facebook.com/story.php?story_fbid=1514898245477805&substory_index=0&id=1514871015480528
Watch Clint's American Ninja Warrior submission video https://www.youtube.com/watch?v=AnYvr493WUY&feature=youtu.be
Don't forget to tune in on Monday, July 10th to watch Clint in action!
- Details
Specialty Support Groups
Caregiver Support Group - Sara Wasserman - (928) 301-8761 - artspeaks10@hotmail.com
Facial Paralysis Support Group - Tara Paul - tnpaul@comcast.net
Headaches (Pre-Treatment) - Ken Posner - (248) 464-0808 - kennyposner@gmail.com
Headaches (Post-Treatment) - Agnes Garino - (314) 821-1905 - algarino@sbcglobal.net
Newly Diagnosed / Pre-Treatment - Chad Nye - (407) 496 - 8357 - chadnye@gmail.com
Young Adults Support Group - Emily Truell - (920) 229-5201 - emily@lacampground.com
Michelle Nemeth - (703) 772-7114 - michelle.f.nemeth@gmail.com
Support Groups by State
We are currently recruiting support group facilitators in the following locations, or contact us today if you would like to start a group in your area.
FL - Ft. Lauderdale
FL - Sarasota
IN - Indianapolis
MA - Boston
MO - Kansas City
PA - Pittsburgh
ALABAMA
MOBILE
Leader: Alan Heffernan
251-621-5665
alanheff@gmail.com
ARIZONA
PHOENIX
Leader: Tracy Denmark Schwimmer
602-561-1855
marcschwim@aol.com
TUCSON
Leader: Tricia Jennings
520-825-9845
azchipster@gmail.com
Leader: Carol Franklin
520-490-5035
lefsequeen53@gmail.com
CALIFORNIA
LOS ANGELES
Leader: Leah Keith
leahkeith@me.com
Leader: Kimberly Ary
kimberly.ary3@gmail.com
PALO ALTO
Leader: Christina Esfehani
650-575-6990
esfehani@yahoo.com
Co-Leader: Mark McLaren
925-838-8175
calpedaler@earthlink.net
SACRAMENTO
For more information, contact our Manager of Volunteer Programs at 770-205-8211 or email volunteers@anausa.org
SAN DIEGO
Leader: Helen McHargue
760-728-0309
foodsmarts@gmail.com
Leader: Margaret Briggs
619-500-6141
glmrchk@gmail.com
Leader: Melissa Diaz
missdiaz@me.com
SAN FRANCISCO-OAKLAND
Leader: Carol Most Walker
415-889-3547
carol@wedrinktea.com
Co-Leader: Thomas Sattler
415-864-0764
tsattler@gmail.com
COLORADO
DENVER
Leader: Ginnie Ferraro
303-469-6391
ginnietennis@hotmail.com
CONNECTICUT
NEW HAVEN
Contact: Kelly Michaud, RN, BSN
kelly.michaud@ynhh.org
For more information, contact our Manager of Volunteer Programs at 770-205-8211 or email volunteers@anausa.org
DC
WASHINGTON, DC
Leader: Allison Shaffer
301-279-9166
akgshaffer@gmail.com
Co-Leader: Michelle Nemeth
703-772-7114
michelle.f.nemeth@gmail.com
FLORIDA
NORTH FLORIDA
Leader: Dyanne Hughes
904-624-0940
dyhughes55@comcast.net
ORLANDO
Leader: Chad Nye
407-496-8357
chadnye@gmail.com
GEORGIA
ATLANTA
Leader: Meredith Daly
678-525-1941
meredith@meredithdaly.com
KENTUCKY
LOUISVILLE
Leader: Kathy Gardner
502-593-9224
kathyc0407@aol.com
LOUISIANA
NEW ORLEANS
Leader: Joan Lemmon
504-261-6490
joanmlemmon@gmail.com
MARYLAND
BALTIMORE
For more information, contact our Manager of Volunteer Programs at 770-205-8211 or email volunteers@anausa.org
MICHIGAN
Leader: Rick May
734-812-8054
rmay726@gmail.com
Meets in W. Bloomfield
MINNESOTA
MINNEAPOLIS-ST. PAUL
Leader: Laura Ferenci
612-501-2248
lkferenci@gmail.com
Leader: Steven Blons
612-925-5926
bodysoul@earthlink.net
MISSOURI
COLUMBIA
Leader: Carol Gardner
573-690-3296
carolhgardner@gmail.com
ST. LOUIS
Leader: Agnes Garino
314-821-1905
algarino@sbcglobal.net
Co-Leader: Phyllis Trulock
314-822-3221
ptru20@yahoo.com
NEW HAMPSHIRE/VERMONT
LEBANON
Contact: Shannon M. Bagley
603-650-8123
shannon.m.bagley@hitchcock.org
For more information, contact our Manager of Volunteer Programs at 770-205-8211 or email volunteers@anausa.org
NEW JERSEY
SOUTH
Leader: Iryna Mosendz
609-705-6623
lutirinas@gmail.com
NEW YORK
ALBANY-MID HUDSON
Leader: Tracey Collins
581-382-3283
Tracey.Collins6273@gmail.com
NEW YORK CITY
Leader: Miranda Warren Sacharin
617-879-0578
m.sacharin@gmail.com
ROCHESTER
Leader: Tom Banach
585-732-4377
tom_banach@hotmail.com
NORTH CAROLINA
Leader: Stewart Binder
919-624-3256
marylandterp.888@gmail.com
Leader: Lisa Fisher
412-996-2386
lfisher@rubollc.com
Meets in Chapel Hill
OHIO
CINCINNATI-DOWNTOWN
Leader: Emily Praeter
513-265-1350
elpraeter@gmail.com
CINCINNATI-WEST CHESTER
Leader: Alice Wedding
513-831-0336
aj2mydad@yahoo.com
NE OHIO
Leader: Pat Gillespie
330-605-7244
cpgillespie@hotmail.com
OREGON
PORTLAND
Leader: David Noyes
503-419-7418
dcnoyes@comcast.net
PENNSYLVANIA
PHILADELPHIA
For more information, contact our Manager of Volunteer Programs at 770-205-8211 or email volunteers@anausa.org
READING
Leader: Nancy Graffius
610-939-9114
ngraffius@comcast.net
RHODE ISLAND (SE NEW ENGLAND)
EAST GREENWICH
Leader: Tony DiBella
401-523-9467
ajd@orgtransitions.com
SOUTH CAROLINA
COLUMBIA
Leader: Ricky Roberson
843-814-2027
rroberson@millsautogroup.com
TENNESSEE
NASHVILLE
Leader: Lauren Vaden
615-232-4929
tarheelbluesy@gmail.com
TEXAS
DALLAS-FT.WORTH
Co-Leader: Linda Dorasami
817-244-0615
anausa.dfw@gmail.com
HOUSTON
Leaders: Shawn and Connie Wilkins
832-458-0236
connie.jo.wilkins@gmail.com
Leader: Mariana Irato
832-474-4090
mariana_irato@yahoo.com
UTAH
SALT LAKE CITY
For more information, contact our Manager of Volunteer Programs at 770-205-8211 or email volunteers@anausa.org
VERMONT/ NEW HAMPSHIRE
LEBANON, NH
Contact: Shannon M. Bagley
603-650-8123
shannon.m.bagley@hitchcock.org
For more information, contact our Manager of Volunteer Programs at 770-205-8211 or email volunteers@anausa.org
VIRGINIA
ROANOKE
Leader: Johnny Diaz
904-738-5063
johnny.diaz@yahoo.com
WASHINGTON
SEATTLE-TACOMA
For more information, contact our Manager of Volunteer Programs at 770-205-8211 or email volunteers@anausa.org
WISCONSIN
GREEN BAY
Leader: Bonnie Simon
920-731-4647
bjs3323@gmail.com
MILWAUKEE
Leader: Judy Jones
414-491-6249
jjonescurran539@gmail.com
- Details
Mark Ruffalo's Story
Award-winning actor and AN patient Mark Ruffalo discusses his acoustic neuroma journey.

































